Estimated reading time: 5 minutes
Central venous catheter-related bloodstream infections are challenging hospital-acquired infections that occur in an estimated 250,000 to 500,000 patients annually in the United States, (1) have a 10%–30% mortality rate, and burden the health
care system with an additional $300 million to $2.3 billion a year. (2). Maintaining high standards for care and maintenance of the dressings, including proper CHG dressing placement, can help prevent catheter-related bloodstream infections at your hospital.
On average, 50-60 patients die daily in US hospitals due to bloodstream infections. (1) In many, if not most, cases, these bloodstream infections are often preventable. The CDC recommends the use of chlorhexidine-impregnated dressings for the prevention of intravascular catheter-related infections. The most commonly used chlorhexidine (CHG) dressing I have seen is the BioPatch® dressing.
How do chlorhexidine (CHG) / BioPatch® dressings work?
One of the critical parts of the central line bundle is antiseptic skin preparation before catheter insertion. This skin preparation, most commonly with CHG, reduces colony counts of bacteria on the skin surface for the central venous catheter insertion for about 48 hours at the most. After that, without prevention, there is recolonization with skin bacteria.
CHG dressings are designed to slowly release CHG to prevent bacterial re-colonization during the 7-day period between dressing changes. (1) BioPatch® specifically is a polyurethane foam disc with chlorhexidine gluconate, or CHG, and is used to combat a wide variety of gram-positive and gram-negative bacteria in patients. If using a CHG disk such as the BioPatch®, the circular foam material provides full circumferential coverage of the insertion site and can absorb and draw fluids away from it.
Replace dressings used on short-term CVC sites at least every seven days for transparent dressings, except in pediatric patients where the risk of dislodging the catheter may outweigh the benefit of changing the dressing [87, 93]. Category IB
What Do You Really Know About the BioPatch®
Most teaching institutions use some form of CHG disc to help prevent catheter-related bloodstream infections. What do you really know about that little disc you place before the final dressing?

Question
Your answer:
Correct answer:
Your Answers
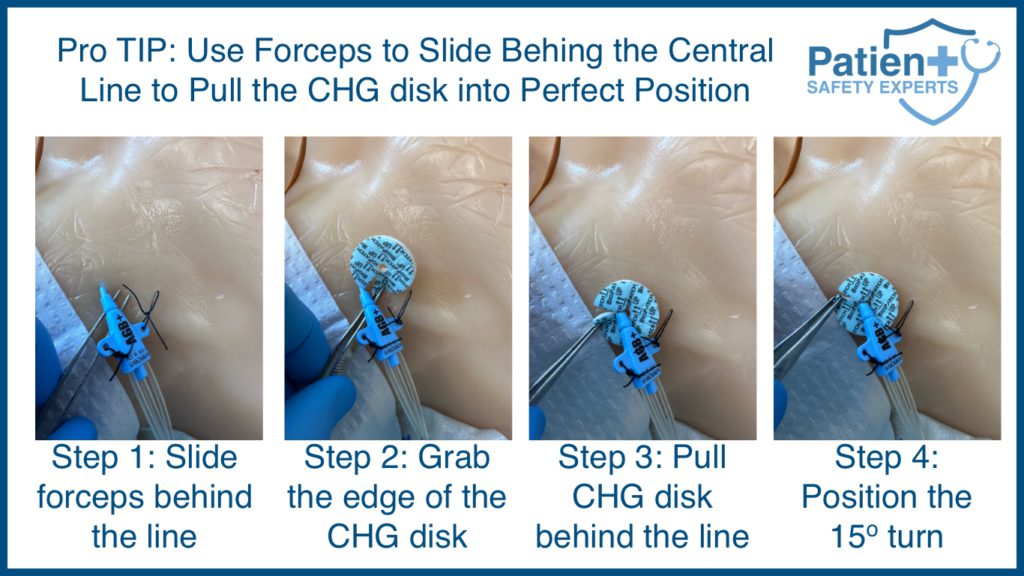
5 Steps to a Proper CHG Disc Placement to Prevent CLABSI
When applying a CHG dressing to a patient, the first thing you will want to do is to prepare the insertion site. Thoroughly cleanse and dry the location you will be applying the dressing. If this is the first dressing, take some time to wipe away excess blood from the insertion site and hold pressure at the insertion site for a few minutes to help subdue the ooze.
Next, you will want to place the circular disc so the slit is either near or underneath the patient’s catheter. You will also want to ensure the slit faces the catheter and then turn it 15-20o in either direction so the catheter no longer sinks through the slit.
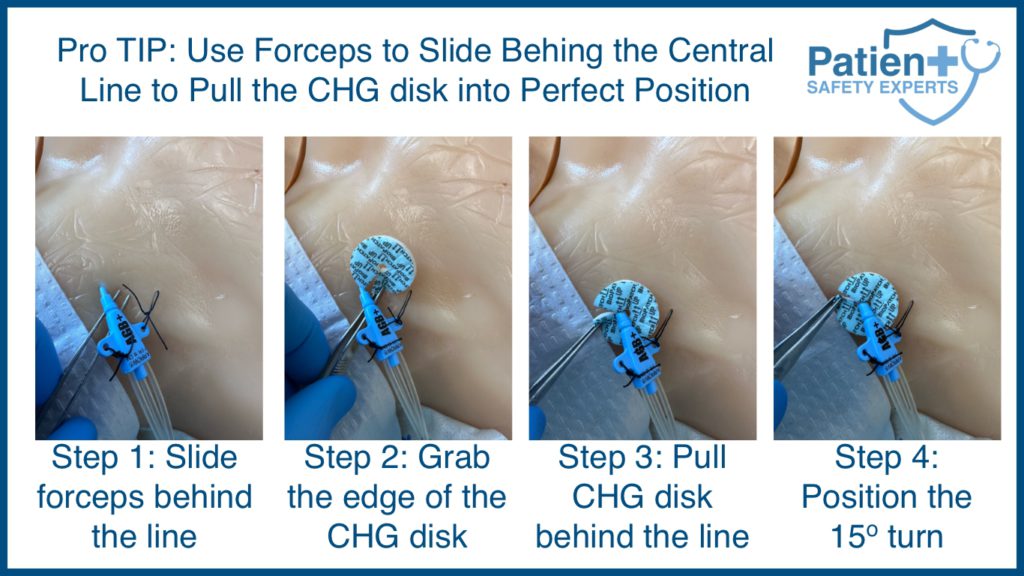
After the turn, It’s essential that the edges of the dressing are together to maximize its effectiveness. When placing the CHG dressing, you should place the dressing with the printed side up for easy removal.
Once the CHG dressing is placed, cover it with an additional bioocclusive dressing and write the date and time on it. CHG dressings will maintain their effectiveness for one week as long as it is kept dry and intact. However, if the dressing becomes saturated with blood or other fluids, it will be necessary to change the entire dressing.
When placed correctly, CHG dressings protect patients from bacteria, microflora, and other organisms on their skin that pose a risk for catheter-associated bloodstream infections.
5 Most Common CHG Disk Placement Mistakes
- Not starting with a clean, dry application site. Make sure all excess blood and fluids are removed before application. If the site is not completely dry, placing the dressing to provide complete 360-degree skin contact around the catheter is difficult to provide maximal efficacy.
- Ensure the catheter has not been secured too close to the insertion point or is “hubbed”. When inserting the central venous catheter, leave about 1 1/2 cm of catheter length out of the skin. The catheter mustn’t be sutured too close to the entry point to allow the width of the CHG dressing to pass behind the catheter. To continue proper skin antisepsis during dressing changes, it is important to pass the CHG dressing behind the line carefully. Do not place the CHG dressing directly onto the catheter; it must be in complete 360-degree skin contact with the skin to be effective.
- Make sure the blank foam side is placed facing down and against the skin. This ensures the foam can slowly release CHG to prevent bacterial re-colonization.
- Do not place CHG dressings directly on or over infected wounds or burns. Please follow local policy regarding these patients.
- Not Replacing the CHG dressing when it becomes saturated with blood or fluids. CHG dressings are designed to absorb some fluids, but once the dressing becomes saturated, it can lose its complete 360-degree contact with the skin and effectiveness.
BioPatch® / CHG Dressing FAQs
1. Can CHG Dressings Be Used On Children & Infants?
No. Do not apply CHG dressing to children or infants. The use of these dressings on premature infants has resulted in hypersensitivity reactions and necrosis of the skin. The safety and effectiveness of CHG antimicrobial dressing have not been established in children under 16.
2. When should I use a BioPatch® or CHG dressings when accessing a port?
Only use a CHG dressing if the patient will have their port accessed for more than 24 hours.
3. Can I use a BioPatch® / CHG dressing if a patient has a skin sensitivity to chlorhexidine swabs?
No. BioPatch® / CHG dressing is a chlorhexidine-impregnated sponge and will likely cause the patient to react adversely. Allergy to chlorhexidine has been increasingly reported, particularly in perioperative and other procedural settings. The hypersensitivity reactions range from mild cutaneous reactions to anaphylaxis or death.
References:
- Veenstra D.L., et al.: Efficacy of antiseptic impregnated central venous catheters in preventing catheter-related bloodstream infection: A meta-analysis. JAMA 281:261–267, Jan. 20, 1999.
- Smith P.W., et al.: Society for Healthcare Epidemiology of America/Association for Professionals in Infection Control and Epidemiology Guideline: Infection prevention and control in the long-term care facility. Am J Infect Control 36:504–535, Sep. 2008.